The concept of biological manufacturing uses living cells as microfactories to produce medicines, antibodies, and vaccines. While the basic premise of biological manufacturing is somewhat like brewing beer, it operates at a much higher technological level of production than brewing. Living cells are used to create the desired product in biological manufacturing; however, this process involves both chemical and biological processes. This represents the core of modern medicine and has created many treatments that were previously considered fictional.
It is the achievement of successful biological manufacturing that presents the real challenges in this field. Unlike in automotive manufacturing (which can produce identical components by replicating the blueprint for one part), scaling strategies for biomanufacturing cannot be replicated in this manner. Biological manufacturing is more akin to baking a perfect cupcake than baking a perfect cake the size of a swimming pool. As the number of “batches” increases, so too does the stress placed on the “chef,” or living cell, ultimately resulting in “bad” batches of product that cost millions and delay patients from obtaining their needed treatment.
Scientists and engineers have made great progress in overcoming the roadblocks to mass-producing biologic drugs by developing new manufacturing methods. Examples of these advancements include scientists using AI to predict optimal environmental conditions for producing their products and creating a continuous “assembly-line” process for producing cell-based products. These advancements have helped many people obtain biologic drugs.
The biggest challenge facing the development of biologic drugs, enzymes, and other cell-based products is converting the small-scale methods used in labs to create these products into commercially viable manufacturing systems. To scale up biomanufacturing successfully, you must consider all three major components of the product cost: product quality, increased production volume, and reduced regulatory hurdles.
First, you need to understand which variables most affect performance. Using a risk assessment approach, you can determine which variables are “scale-sensitive” in the bioprocess (i.e., mixing, gas exchange, shear force, thermal control, and hold time). Biomanufacturing companies will use validated laboratory models to test design changes before building larger tanks; this provides an opportunity to evaluate design alternatives quickly and at low cost.
Scale up for stability and robustness, not maximum yield. Develop and document a proven operating range of key variables, including pH, temperature, feeding schedules, agitation speed, etc. Also, scaling up biomanufacturing processes that use continuous or real-time control to improve product uniformity across batches will facilitate technology transfer within different locations and/or partner companies.
Scale-up planning is most effective when both the upstream and downstream processes are planned simultaneously. If the titer from the upstream process is too high, it may exceed the capacity of downstream processes (e.g., filtration, chromatography) or overload the preparation of buffers required for downstream processing. Therefore, consider opportunities for debottlenecking from the outset of scale-up planning using techniques such as single-use flowpaths, intensified/continuous processing strategies, optimized column sizing, and/or improved scheduling of shared utility systems.
There are going to be many good opportunities for the development of scalable biomanufacturing strategies by considering the biomanufacturing facility as an integrated system (i.e., not as separate units) and through digital tools (e.g., advanced analytical tools; automation) that reduce variability due to the potential for human error and increase the rate at which new knowledge is applied to implementation.
Several companies in the industry have already implemented Process Analytical Technology (PAT), Electronic Batch Records (EBRs), and Model-Based Control (MBC). The adoption of PAT, EBRs, MBC, and other related technologies and scalable strategies for biomanufacturing will provide the opportunity to greatly enhance the ability to identify and correct deviations and quickly conduct investigations when they do occur.
Scaling strategies for biomanufacturing should also consider your supply chain and workforce. Prior to initiating the scale-up, ensure that you have secured all necessary raw materials, identified and qualified alternative suppliers, trained all personnel using standardized procedures on all shifts, and documented the rationale and validation logic used during execution of the scale-up activities. By implementing scalable biomanufacturing strategies that integrate the principles of science, engineering, quality, and operations, you will be able to reliably scale up biomanufacturing processes with each successive batch.
#Healthcare AI Tools: Transforming Hospital Operations
Why Making More Isn’t as Simple as Multiplying the Recipe
While anyone who cooks can tell you that doubling or tripling the ingredients on a cake recipe won’t give you a better cake, it’s the same issue with the production of bioproducts in a manufacturing setting. While a failed cake might cost a lot of money and possibly harm people, a failed process at a commercial scale could have catastrophic results.
The problem boils down to the recipe itself. The conditions required to sustain cell health and produce a product in a lab setting do not necessarily equate to the conditions in a larger manufacturing setting.
To solve this problem, manufacturers use a bioreactor. A bioreactor is a large, highly controlled, sterile tank that provides ideal conditions for cell growth. Bioreactors come in all shapes and sizes (from a couple of liters to the size of a small school bus). Manufacturers can also use bioreactors to monitor and regulate factors such as pH and nutrient levels within the vessel. Bioreactors are not merely large mixing bowls; they act as sophisticated life support systems for billions of tiny biological factories.
Although it is important to control the bioreactor environment as a whole, it is very challenging to ensure that every single cell has an appropriate amount of oxygen. Inadequate mixing within the bioreactor can result in “cold-spots,” with low oxygen levels, which can lead to the buildup of potentially toxic waste products elsewhere in the bioreactor. Cold spots and the development of toxic waste products represent significant barriers to the scale-up of bioprocessing operations.
These issues create cellular stress. Cellular stress occurs when cells have insufficient oxygen or nutrients or are exposed to toxins. Cells experiencing cellular stress cease production of the product (e.g., a commercially valuable drug or vaccine) and/or die, resulting in a loss of a batch with potential value of tens of millions of dollars. Therefore, the selection of the cell culture vessel is one of the most critical decisions in developing a viable manufacturing process.
Biomanufacturing Processes – Core Upstream and Downstream Workflows Supporting Large-Scale Production
Biomanufacturing is required for commercial-scale biologic products, as with all other large-scale manufacturing processes; however, it adds complexity to both upstream cell culture and downstream purification, quality, and regulatory compliance. To ensure biomanufacturing processes are reproducible, measurable, and exhibit low variability, even minor variations may result in differences in yield, purity, and critical quality attributes of the final product. Therefore, as volume, equipment size, and throughput increase in the biomanufacturing process, there should be no reduction in performance.
The upstream phase of the biomanufacturing process begins with cell line development and banking. Following this, the cell expansion process increases the seed train to produce a viable, consistent inoculum. When producing on larger bioreactor vessels, teams must control mixing, oxygen transfer, CO2 removal, and shear to maintain cell viability and productivity. Typically, successful scaling strategies for biomanufacturing require small-scale models or process characterization studies to determine how each of the previously listed parameters behaves at larger scales.
Biomanufacturing processes typically consist of several downstream operations, including harvesting and clarification (for example, centrifugation and/or depth filtration), capture chromatography, purification steps, and virus removal. In addition to the other downstream processing steps, ultrafiltration/diafiltration is performed to concentrate the final product and prepare it for formulation by exchanging buffers. Scaling strategies for Biomanufacturing processes can be optimized by balancing the number of downstream operations per unit of upstream product output. The higher the titer, the greater the potential bottlenecks in filter capacity, resin lifetime, and buffer logistics. Methods of scaling up biomanufacturing include: debottlenecking with the correct column size, optimizing cycle timing, and using a single-use flow path when possible.
Both biomanufacturing processes and analytical/quality systems are integral to biomanufacturing. Analytical equipment (e.g., in-process testing, PAT when possible, and trend analysis of deviations) helps detect process changes early. Automation, electronic batch records, and standardization of sampling procedures help reduce human-caused errors and expedite the investigation into the problem’s cause. The same principles used to develop the scaling strategies for biomanufacturing processes are applied to raw materials, suppliers, and employee training to maintain consistent process execution during expansion of the manufacturing facility.
By integrating biomanufacturing processes into a single, cohesive system, scaling shifts from an art of trial and error to a science-based, controlled, and data-driven approach to increasing production levels while maintaining quality.
Upstream vs Downstream Biomanufacturing Workflow (Process Map)
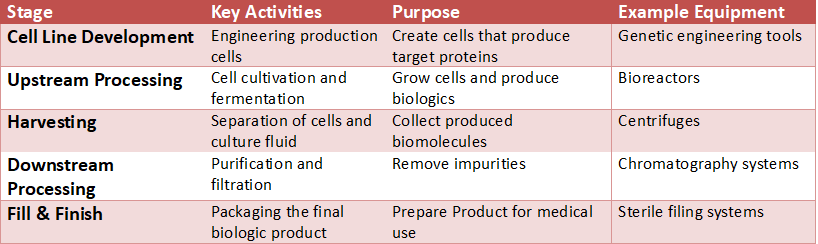
Source: FDA Biomanufacturing Process Overview
https://www.fda.gov/vaccines-blood-biologics/biologics-manufacturing
Biomanufacturing Methods – Proven Techniques for Reliable and Efficient Biomanufacturing Scale-Up
The success of reliable scale-up in biomanufacturing depends heavily on identifying methods that deliver consistent product quality as production volumes increase and costs decrease. Therefore, the most successful Biomanufacturing Methods are those that have been validated, can be transferred from one location to another, and have clearly defined control strategies. Thus, it is advisable to develop your scale-up strategies for biomanufacturing at the same time you select your first method, rather than developing them after you encounter problems with the selected method.
Methods for Biomanufacturing Upstream Production will usually include established, robust cell banking, consistent seed train expansion, and well-defined media and feed programs as the base methods. In scale-up, the aforementioned processes typically utilize both geometric and process-based parameters (e.g., power input, tip speed, oxygen transfer rate) to ensure that growth and productivity rates are consistent across all production scales.
Scaling Strategies for Biomanufacturing can be applied using scale-down models that simulate conditions in large reactors. This allows engineers to quickly test the impact of agitator speed, aeration, and feeding, and assess potential impacts on the process before a full-scale batch is lost. Scale-down modeling is complemented with Biomanufacturing Methods that are centered around monitoring, such as online sensors, PAT, where possible, and tightly controlling pH, temperature, and dissolved oxygen levels to minimize variability.
Biomanufacturing Methods used for downstream clarification aim to clarify the solution while maximizing recovery. The choice of an appropriate clarification technique (e.g., centrifugation, depth filtration) depends on the expected solids concentrations and volumes.
Chromatography remains a cornerstone of purification; however, several key strategies for successful scaling of Biomanufacturing Methods to reduce cycle times and variability include optimal residence time, standardized column packing (using pre-packed columns), and life-cycle management of resin performance. Planning ahead for additional capacity will ensure that increases in upstream titer do not place undue stress on filtration, buffer preparation, and column usage.
In addition to process intensification and modular design as methods for scaling in Biomanufacturing, single-use technologies can be used to decrease the time required to change over equipment and clean equipment. Hybrid systems can also be used by combining disposable flow paths with traditional stainless steel systems to add cost flexibility.
Continuous or semi-continuous processes (e.g., perfusion upstream and continuous chromatography downstream)are another scaling strategies for Biomanufacturing Method, which may allow an organization to produce more product than before without having to build larger tanks. However, these types of systems require close monitoring of all aspects of the system and an effective data system.
Standardized procedures are another Biomanufacturing Method. Documented standard operating procedures, standardized training of personnel, electronic batch records, and deviation trending are all methods that will help to assure consistent performance throughout the day/week/month/site. By establishing clear standardization and scaling strategies for biomanufacturing, organizations will experience improved predictability, efficiency, and compliance as they transition their product from the pilot phase to commercial manufacturing.
Advanced Biomanufacturing – Next-Generation Technologies Enabling Scalable Biomanufacturing Operations

Advanced Biomanufacturing will fundamentally alter how we manufacture biologics and other bio-products as they progress from research and development through to commercial production. Through the use of automation, flexible systems, and “big” data, Advanced Biomanufacturing will enable manufacturers to produce much larger quantities of their products while maintaining product quality. Most teams have two primary goals: to scale up more quickly and predictably, and to do so at a lower cost than they are today. To meet these objectives, Advanced scaling strategies for Biomanufacturing need clear, efficient methods supported by effective and efficient approaches.
Process Intensification (PI) is a key driver for Advanced Biomanufacturing. With upstream perfusion and high-cell-density cultures, Advanced Biomanufacturing can increase cell productivity within a given footprint. Therefore, there is no need for very large bioreactors. In addition, PI will require close monitoring and control, including Advanced Sensors, Automated Feeding Systems, and Model-Based Control Loops. When done properly, Advanced Biomanufacturing will provide a means to create more consistent scaling methods and more predictable transitions across scales and sites.
The Downstream process is also experiencing rapid growth in Advanced Biomanufacturing, including continuous and semi-continuous purification options. These include multi-column chromatography, inline dilution, and closed single-use fluid handling, which all contribute to lowering hold times, decreasing the risk of contamination, and increasing production throughput. It is expected that downstream may become the limiting step as upstream titers continue to grow; therefore, Advanced Biomanufacturing will provide additional support for biomanufacturing scale-up through increased cycle efficiency, enhanced capacity planning, and reduced changeover time.
Digitalization has two pillars: Advanced Biomanufacturing and Real-Time Data Capture. The combination of real-time data capture and advanced analytics enables teams to monitor product quality using process parameters and quickly identify operational deviations. Many organizations are now combining Advanced Biomanufacturing with Process Analytical Technology (PAT), which includes electronic batch records and automated deviation workflow systems.
These technologies have been shown to support scaled-up strategies for biomanufacturing by shortening investigation time, enabling more thorough comparisons during technology transfer, and extending the learning cycle.
The biomanufacturing process is being changed in relation to facility design. Advanced Biomanufacturing, in particular, is moving away from fixed-facility designs (which have historically been used) and toward modular, hybrid, or single-use facility designs.
Single-use facility designs can be used to manufacture many different products within the same facility space. This flexible facility design is enabling reduced capital expenditures to support multi-product manufacturing, and, essentially, Advanced Biomanufacturing is allowing the integration of “modules” to add new products while continuing to operate existing plants, which aligns well with the current scaling strategies for biomanufacturing.
In summary, Advanced Biomanufacturing is utilizing intensified processing, continuous processing, automation, and digital control systems to reduce reliance on trial and error and increase consistency and repeatability when scaling processes — to enable scaled biomanufacturing solutions that provide timely results and are more resilient to meet the needs of future generations of product pipelines.
The Ultimate Choice: A Reusable Steel Kitchen vs. A High-Tech Disposable Bag
The size of your bioreactor is a critical first decision for a bio/pharmaceutical company’s manufacturing process. The next important decision appears much easier: should I build a steel tank that will last for many years and can be cleaned and reused, or should I use a sterile plastic bag?
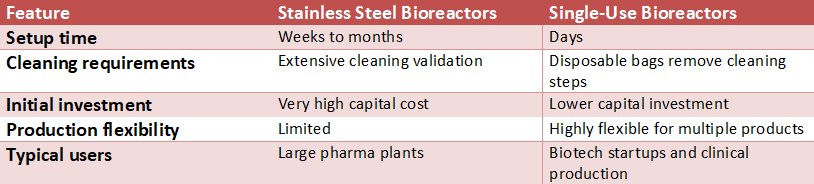
In the past, stainless steel tanks were the norm in this industry – just like those huge pots in a commercial kitchen. They have always been long-lasting, reliable, and reusable for years. Single-use bioreactors (also known as single-use bags) are a new, more versatile technology that has revolutionized the industry since their introduction in the last decade. These are large, advanced, pre-sterilized plastic bags located within a frame.
Single-use technology eliminated tank cleaning. After each batch is finished in a stainless steel tank, the tank must undergo sterilization, which can take anywhere from several days to several weeks before it is ready for the next batch. Contamination is another major concern when the cleaning and sterilizing process is improperly performed.
The risk of cross-contamination is extreme if the cleaning and sanitizing aren’t performed properly. Single-use technology has several advantages: it removes all those risks because once you are done with the bag, you dispose of it and replace it with a brand-new one. Therefore, if your goal is to produce a vaccine today, and tomorrow you want to start making a cancer therapy, there’s no chance of contamination or carryover.
Will the bag always be ideal? No, because that decision will depend on the goals of your mission. Companies have to consider both the advantages (e.g., volume) and the disadvantages (e.g., flexibility) of each option listed above.
- Stainless Steel: The best choice for making large quantities of a single high-volume product (such as insulin) in a continuous manner.
- Single Use: The best option for companies with many different products, each made in small batch quantities, or companies that want to get their products into clinical trials as quickly as possible
Ultimately, the type of container used will influence how the “cooking” process is accomplished. Single-use technology offers two major advantages over stainless steel containers: no cleaning time is required after use, and the production process can be completed at a higher speed and efficiency.

Stainless Steel vs Single-Use Bioreactors
Example: Single-use systems can reduce facility costs by up to 40%
Source: BioProcess International
https://bioprocessintl.com
Moving Past the ‘Bake and Wait’ Model: The Power of Continuous Production
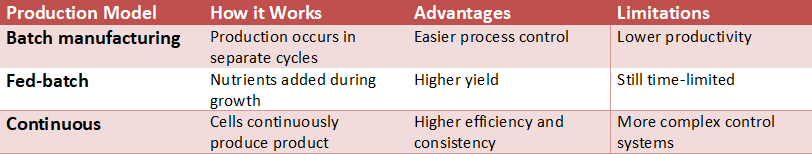
Traditionally, producing biotherapeutics was similar to baking: a large tank (bioreactor) containing all the required ingredients (cells, broth) would be filled and incubated for several weeks. The traditional method of producing a large quantity of product at one time in a single tank (batch production) is an all or nothing risk; the batch will either contain the product you are looking for or the batch will fail due to some type of event, such as contamination or a slight temperature fluctuation, resulting in the loss of the entire batch which could cost millions of dollars.
Continuous biomanufacturing is replacing the traditional batch production model currently used in the manufacture of biotherapeutic products. Continuous biomanufacturing maintains a continuous flow of product throughout the manufacturing process rather than creating batches. Rather than filling a tank with all the required ingredients (raw materials) and allowing it to incubate for several weeks, a small continuous flow of raw materials will enter the bioreactor; the cells will continue to produce the drug, and a purified product will exit the bioreactor continuously.
The transition to this form of manufacturing necessitates the development of a continuous, robust biologics manufacturing process rather than a sequence of separate manufacturing steps.
The many advantages of the new model have the potential to be significant. First, since the equipment size will be significantly reduced and the equipment will operate continuously, facilities will be able to manufacture the same amount of medication in much less space than before. Second, and more importantly, the continuous manufacturing process can significantly reduce risk. If an issue occurs during a continuous process, it will spoil only 2 hours of production instead of a month. Third, the continuous production of the drug substance also enhances and simplifies the last major purification process, which is the main constraint on Downstream Processing.
It should go without saying that the continuous operation of micro-scale manufacturing processes that cannot be shut off will require an extended level of monitoring. You can no longer simply take a sample from the product when the process is finished. Rather, you need to monitor the overall health and the productivity of these ‘living’ factories in “real time” while the process is running. However, how do you monitor one trillion cells within a vessel without opening the vessel or door?
Traditional Batch vs Continuous Biomanufacturing
Example: Continuous biomanufacturing can increase productivity by 2-5x compared with traditional batch processes.
Source: MIT Center for Biomedical Innovation
https://cbi.mit.edu
How Do You Check on a Trillion Cells? With Smart Sensors and Real-Time Data
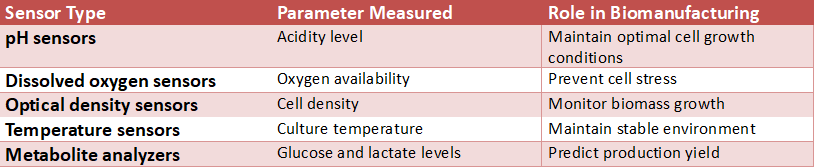
The key to a successful cell-culture product is Process Analytical Technology (PAT), which uses bioreactors equipped with advanced internal sensors to measure, in real time, how well cells perform. The sensors function as a type of “personal trainer” or “doctor” within the bioreactor providing an advanced “fitness tracker” which measures the culture’s temperature, and an “inventory tracker” which measures the level of nutrients available, and the levels of waste products produced by the cells; both types of information go back to the bioreactor’s management team in real time allowing them to be able to monitor if they need to make adjustments to the bioreactor to maintain an optimal condition.
In stark contrast to the traditional “bake and wait” type of production methods, in which a problem was unknown until it was detected through sampling and testing for the problem after the fact, a team utilizing PAT will know in real time that its cells are stressed because there is a lack of nutrients, in addition to many other reasons. This is similar to receiving a dashboard warning light on your car indicating low engine oil, rather than having a mechanic tell you why your car stopped running after the fact. Through this real-time feedback loop, the process may continue without interruption.
Rapid Response capabilities for the active management of a bioprocess are an important component of a broader manufacturing philosophy called Quality by Design (QbD). QbD is based upon the belief that quality should be built into each step of the manufacturing process from the beginning; therefore, instead of waiting until the end of the process and hoping that there will be a good outcome, you build a system in such a manner that an undesirable outcome is virtually impossible.
PAT provides the eyes and ears for QbD. Wouldn’t it be wonderful if PAT could provide the ability not only to monitor the current state of your cells, but also predict how they may behave in the future?

AI and Smart Sensor Monitoring in Bioreactors
Example: Real-time bioprocess monitoring systems allow predictive control of cell cultures, improving yields by up to 20-30%.
Source: Nature Biotechnology – Bioprocess Monitoring Research
https://www.nature.com/nbt
Building a ‘Flight Simulator’ for a Bioreactor: How AI Predicts the Future
The data flows continuously from smart sensors, which are more than just medical information; they represent the power source for artificial intelligence (AI) to drive decisions. Researchers create a digital “twin” of the bioreactor by streaming real-time, continuous data to the AI. This allows the researcher to run through multiple “what if” scenarios with the virtual bioreactor, simulating various types of failures without wasting any materials or living cells.
Researchers then replicate those conditions on a much smaller scale, creating miniature versions of the bioreactors in the lab to ensure accuracy. Researchers use a similar method of testing recipes on a small scale in a controlled mini-environment to assess cell limits and observe how cells react under extreme conditions. The research findings provide valuable insights into the behavior of the full-scale system and enable the AI to learn to predict its own performance under all potential circumstances—good or bad.
With both components working together, the AI becomes a reliable predictive tool. The AI can examine all variables affecting the bioreactor’s operation to find the best “recipe” to optimize performance. It can also serve as a prognostic tool to detect early warning signs of trouble before they occur in the actual production process. The ability to have a “crystal ball” view of potential problems allows for cost savings from fewer mistakes and increases the odds of success with each batch produced. However, what happens when there is no need for millions of doses and the “batch” is created for only one individual?
Global Biomanufacturing Market Growth
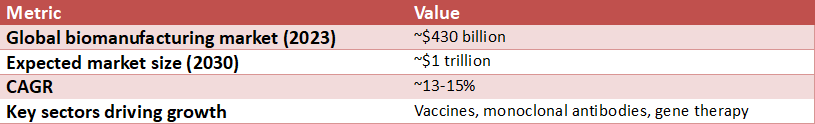
Source: McKinsey Biomanufacturing Report
https://www.mckinsey.com/industries/life-sciences
Biomanufacturing Efficiency – Improving Yield, Speed, and Cost-Effectiveness at Scale
Biomanufacturing efficiency is defined as the ability to manufacture products at the highest possible yield (amount), while keeping costs as low as possible and cycle time as short as possible. Efficiency in large-scale biomanufacturing is determined by many small, independent decisions made throughout all four phases of the biomanufacturing process: upstream, downstream, analytical, and operational. This is why good scale-up strategies for biomanufacturing can serve as a framework for relating these decision-making steps, so that an improvement in one phase does not create a bottleneck in another.
The typical way to achieve upstream improvements in biomanufacturing efficiency is to start with a cell line that produces a constant number of cells to seed cultures consistently, and to use the best available media and feed conditions. By controlling key variables, including dissolved oxygen levels, pH, temperature, and feeding conditions, you can reduce cellular stress during the manufacturing process, thereby increasing productivity.
Biomanufacturing efficiencies were increased by using scale-down models to rapidly evaluate upstream process changes and identify “acceptable ranges” for all parameters, thereby providing a robust process. These two approaches have been at the heart of developing effective scaling strategies for biomanufacturing, reducing batch failures, and facilitating technology transfer from development to production.
In addition, the efficiency of the downstream portion of the biomanufacturing process depends on both the volume of product generated per unit time and the percentage of product recovered. Therefore, when designing systems for clarification, filtration, and chromatography, the size of the equipment must be based on expected product volumes and titers, as well as reasonable estimates of the time required for each operation and the potential lifespan of the resin(s).
Biomanufacturing efficiency can also be enhanced by optimizing buffer usage and reducing the time products are held, while maintaining the same purification train.
The strategies of identifying potential bottlenecks in downstream biomanufacturing, scheduling intelligently, and selecting formats (single-use or stainless steel) that meet the needs of the company’s product portfolio and changeover needs are key to efficient scaling strategies for biomanufacturing process into downstream
Data and automation are both major drivers of scaling strategies for biomanufacturing. Teams that utilize real-time monitoring, electronic batch records, and trend analysis of alert signals achieve better outcomes than those that do not. The analytics (which tie together the Process Parameters to the Critical Quality Attribute(s) [CQAs] of the product) will assist with identifying the most significant changes for optimization.
Tools that assist with scalable biomanufacturing strategies will increase process knowledge, improve comparability, and reduce the time required for investigations.
Equally important to tools for improving Biomanufacturing Efficiency are operational disciplines. Improving Biomanufacturing Efficiency by minimizing human-related errors and reducing rework time can be achieved by implementing standard work, providing proper education/training, and ensuring clear material-flow processes.
Protecting Biomanufacturing Efficiency through Supply Chain Planning (including raw material qualification, alternative sources, and inventory management strategy) will minimize/eliminate downtime due to shortages and/or delayed quality releases.
Biomanufacturing Efficiency at a larger scale ultimately depends on striking a balance among three key areas: Science, Equipment, and People/Data. Organizations that approach Biomanufacturing Efficiency as a total-systems goal and consistently use their own scalable biomanufacturing strategies will achieve higher yields, faster production, and lower costs while maintaining product quality.
Sustainable Biomanufacturing – Eco-Friendly Approaches for Long-Term Biomanufacturing Growth

Sustainable Biomanufacturing is characterized by creating products and processes that minimize their negative impact on the environment without compromising product quality or availability through reliable production methods, or without a strong financial basis. Sustainability within Commercial Scale Biomanufacturing involves multiple choices regarding both design and operation during product manufacture to reduce the overall amounts of lifecycle energy required, water consumed, waste generated, and emissions produced by these activities.
Linking a facility’s sustainability goal to its scaling strategies for biomanufacturing is the most effective way to ensure growth does not increase its footprint.
In addition to the benefits of improved heat transfer, heating/ventilation/air conditioning (HVAC), and cleanroom classification, energy-efficient utility systems may provide substantial reductions in power usage. Furthermore, water-reduction strategies, including CIP (clean-in-place) schedule optimization, using reusable water where possible, and scheduling techniques to optimize resource utilization, will help create sustainable biomanufacturing processes while reducing operating costs.
These areas will provide ways to scale up biomanufacturing strategies by expanding the facility’s capabilities while remaining within the constraints of the existing site utilities.
Additionally, the concept of Sustainable Biomanufacturing will be enhanced by using materials and consumables that support sustainability. This may include using high-concentration buffers in smaller volumes to reduce single-use plastics whenever possible, and purchasing all products and materials used in the facility from suppliers that meet the facility’s requirements for low carbon content.
In addition to enhancing sustainability, single-use systems in sustainable biomanufacturing will help reduce the use of cleaning agents, water consumption, and downtime, particularly in multi-product manufacturing environments. Therefore, it is critical to determine which strategy(s) best achieve sustainability from a life-cycle perspective and to develop them into scaling strategies for biomanufacturing from the very beginning of product development.
Sustainability in the context of scaling strategies for biomanufacturing (Sustainable Biomanufacturing) involves a variety of activities. These include reducing the amount of consumables required for production, the number of batches produced before failure, and the energy consumed per unit of product produced. It also includes digital monitoring, automation, and control of processes that minimize deviations from normal operation, thereby reducing the need for rework, reducing the risk to the sustainability of Sustainable Biomanufacturing, and improving the facility’s operational reliability. Therefore, these strategies provide practical means to scale up biomanufacturing while maintaining product quality.
In addition to achieving a sustainable process, Sustainable Biomanufacturing also requires an emphasis on waste recovery and circularity. Sustainable Biomanufacturing can involve the development of segregation and recycling programs; the optimization of solvent and chemical use; and partnerships with organizations that enable the recovery or environmentally responsible disposal of waste materials. Teams developing Sustainable Biomanufacturing processes can also consider implementing alternative packaging and logistics solutions to reduce greenhouse gas emissions associated with transporting materials and products.
To use Sustainable Biomanufacturing as a continuous process (and not just an event), you need to identify how your current production process is impacting the environment and set specific goals.
In addition, when you create long-term sustainability by implementing a leaner, more efficient process that reduces your resource use, it will help mitigate your exposure to factors beyond your control, such as variable energy costs, variable water availability, and regulatory changes.
When you develop scale-up plans for your biomanufacturing based on sustainable biomanufacturing principles, you can increase your bioproduction capacity while continuing to reduce your environmental footprint without compromising your product quality or supply.
The New Frontier: Scaling for Personalized Cures Like Cell and Gene Therapy
The final question—how to make a single batch of medication for one person—underlies the most forward-thinking treatments now being developed, including cell and gene therapy. These treatments use a patient’s own living cells to create their “treatment,” thus making it uniquely tailored to that patient. Thus, this reverses the typical bioprocessing challenge (i.e., scale up a single formula in a large bioreactor) and will require “scale out” or safely and accurately producing hundreds of different, individualized batches of product side-by-side in parallel.
This is the difference between baking one gigantic cake that fits on a car and running a bakery that bakes 100 different custom birthday cakes.
Beginning to conceptualize the vast complexity of creating an innovative biomanufacturing method for personalizing medicine requires integrating all the breakthroughs mentioned earlier. The single-use bioreactors will offer an additional level of protection against contamination by using less than 10 liters per product batch. In addition, the “digital twin” technology will be able to monitor every single patient-specific batch produced on a “real-time” basis. Taken together, this represents a new paradigm that should allow the highest possible degree of confidence in producing high-quality biologic products for each unique patient treatment, something that could never be achieved solely through human effort.
To summarize, these new approaches for scaling up in the production of biologics are providing the foundation for the development of a new type of factory; rather than a factory designed to optimize production of a single best-selling drug, it has the flexibility to be utilized as a generic manufacturing platform to produce individualized treatments for patients. The technologies that enable mass vaccination are the same ones that could drive the future of personalized treatments. Biomanufacturing is transitioning a previously artistic process into a science.
Biomanufacturing Trends – Emerging Innovations Shaping the Future of Biomanufacturing

Biomanufacturing Trends are transforming the industry by creating an environment that is less dependent on “large batch” and is a smarter, faster, and more adaptable method of production. A number of modern biomanufacturing trends focus on reducing product development timelines, improving product robustness, and creating a reliable supply of products with differing characteristics (i.e., complex modalities). Additionally, Biomanufacturing Trends will define parameters for future capacity planning and scaling to establish the commercialization requirements for large-scale manufacturing.
Two major Biomanufacturing Trends are process intensification and the increased use of continuous and semi-continuous downstream processing. In recent years, there has been increased emphasis on high-cell-density and perfusion-based processes, as these methods enable manufacturers to produce at significantly higher yields without increasing facility footprint. The implementation of process intensification within biomanufacturing allows manufacturers to increase capacity while utilizing existing facilities; this is achieved by increasing productivity rather than increasing the size of production equipment. Similarly, continuous and semi-continuous downstream processing methods are becoming increasingly popular as a means of increasing the rate of product flow through the manufacturing process, reducing holding times, and providing a smooth transition between individual unit operations.
Single-use and hybrid manufacturing units remain among the most popular biomanufacturing trends for multi-product manufacturers. Disposable flow paths reduce the need for cleaning, shorten changeover times, and enable quicker setup of new lines. A combination of the two, hybrid manufacturing units, utilizes the benefits of disposable manufacturing while still utilizing the durability of stainless steel when high volumes are produced.
Modular growth through single-use and hybrid biomanufacturing trends supports scalability by enabling easy expansion and reducing downtime during growth. The frequency of biomanufacturing trends, related to digitalization, continues to increase across the entire value chain of the biopharmaceutical industry. The use of sensors and Process Analytical Technology (PAT), and associated systems, such as data historians, electronic batch records, etc., has allowed for better visibility into processes, increased control over them, and a way to correlate process parameters to product quality, and to recognize trends or “drift” which could indicate an issue in a process.
Thus, the application of digitalization technologies and analytics can enable scaling strategies for biomanufacturing space by promoting data commonality, enabling quicker root cause identification, and reducing the number of deviation events resulting from higher production volume.
The second area of Biomanufacturing Trends is the workforce and operational aspects of biomanufacturing: standardization, automation, and right-first-time (RFT) manufacturing execution. With the continued diversity of products entering pipelines and supply chain unreliability stemming from various factors, companies have been developing ways to improve operational resiliency through alternative sourcing, improved raw-material qualification, and risk-based validation. The operational Biomanufacturing Trends identified here provide companies with the means to maintain reliable supply options as production volumes increase.
The third area of Biomanufacturing Trends includes sustainability. Sustainability was once viewed as a desirable business practice, but is now an expected one. Examples of sustainability practices include optimizing energy use, conserving water, and considering the life cycle of the materials you use. Companies that successfully deploy new and innovative products and processes through discipline-based, scalable strategies for biomanufacturing will likely experience faster, more flexible, and more sustainable growth than those that do not.
From Art to Science: The Future of Biomanufacturing
Biological advancements have allowed what was once unattainable (the ability to scale up the complexity of biological processes) to become a predictive science. As we continue to develop a systematic method to translate the fragile biology recipe into a predictable manufacturing process – from a black box to a solvable problem using an evolving engineering architecture – we continue to make progress on this path.
Advances are being achieved through new technologies that transform uncertainty into reliability. Flexible, single-use manufacturing facilities operate similarly to a workshop; Continuous Manufacturing is a continuous production line for molecules; Smart Sensors monitor the living cells continuously; Artificial Intelligence functions as a “crystal ball” for scientists to predict possible outcomes and anticipate results. Collectively, these technologies represent a robust suite for expanding our understanding of the biological world – ensuring that successful methodologies developed within Laboratory Environments can be utilized globally.
Biomanufacturing represents the next step beyond medicine. The same approaches will be foundational to sustainable food production without harming the environment, the development of biodegradable plastics, and the production of clean biofuels. A quiet revolution is taking place – to domesticate the microscopic world to address our greatest macroscopic problems.